cbx2在青鳉胚胎发育和性腺中的表达谱与定位
Expression pattern and localization analysis of cbx2 during embryonic and gonadal development in the medaka (Oryzias latipes)
-
摘要: CBX2(Chromobox homolog 2)作为PcG(Polycomb Group)蛋白家族成员之一,在性腺发育中具有重要作用。缺乏cbx2的小鼠,雌雄性腺均发育不良,并且部分小鼠出现雄性转变为雌性的性反转现象。实验选择模式生物青鳉作为对象,通过荧光定量PCR和免疫组化技术检测了cbx2在青鳉胚胎发育过程和性腺中的表达。定量结果显示,在胚胎发育时期,cbx2在原肠胚期、神经胚期和器官形成期均具有高的表达;在两性性腺中,cbx2在精巢中的表达量较高。免疫组化结果表明,在精巢中,CBX2主要在精原细胞和精母细胞中表达;在卵巢中,CBX2主要在Ⅰ、Ⅱ、Ⅲ时相卵母细胞中表达。为进一步研究cbx2的功能,实验利用显微注射技术对cbx2进行RNA干扰,结果显示,注射siRNA后,随着cbx2表达量降低,雄性性别相关基因sox9(SRY-related HMG box 9)的表达也相应下调,雌性性别相关基因foxl2(Forkhead transcriptional factor 2)的表达则上调。研究表明,cbx2不仅参与了青鳉胚胎发育过程,而且在青鳉性腺分化以及雌雄配子发生中也起重要作用。
-
关键词:
- 青鳉 /
- 胚胎发育 /
- 性腺发育 /
- cbx2" type="keywords.keywordCn" href="javascript:void(0);"> cbx2
Abstract: CBX2 (Chromobox homolog2), as a key member of the Polycomb Group protein (PcG) family, plays an important role in gonadal development. Mice lacking cbx2 showed defects in gonads (both ovary and testis) development, and some mice have exhibited male-to-female sex reversal. But little is known about the function of cbx2 in gonadal development of fish. In this study, we used Oryzias latipes, a model organism, to investigate the expression of cbx2 during embryonic and gonadal development by real-time PCR and immunohistochemistry. Real-time PCR results showed that cbx2 was highly expressed in embryos at gastrula, neurula and organogenesis stages. In bisexual gonads, cbx2 had a relatively high expression in testis. Immunohistochemistry results revealed that cbx2 mRNA was mainly localized in spermatogonia and spermatocytes in testis. And cbx2 was also predominately observed in oocytes at stages I, II and III in ovary. In order to further study the function of cbx2, we used microinjection technology to knock out cbx2. The results showed that the expression of cbx2 was significantly decreased after injecting siRNA, and thus the expression of sox9 (SRY-related HMG box 9) was down-regulated, whereas the expression of foxl2 (Forkhead transcriptional factor 2) increased. This indicated that cbx2 was not only involved in embryonic development but also played a role in gonadal differentiation and gametogenesis in O. latipes.-
Key words:
- Oryzias latipes" type="keywords.keywordEn" href="javascript:void(0);"> Oryzias latipes /
- embryonic development /
- gonadal development /
- cbx2" type="keywords.keywordEn" href="javascript:void(0);"> cbx2
-

-

图 图版Ⅰ 青鳉胚胎不同发育时期
Figure 图版Ⅰ. Different developmental stages of O. latipes embryos
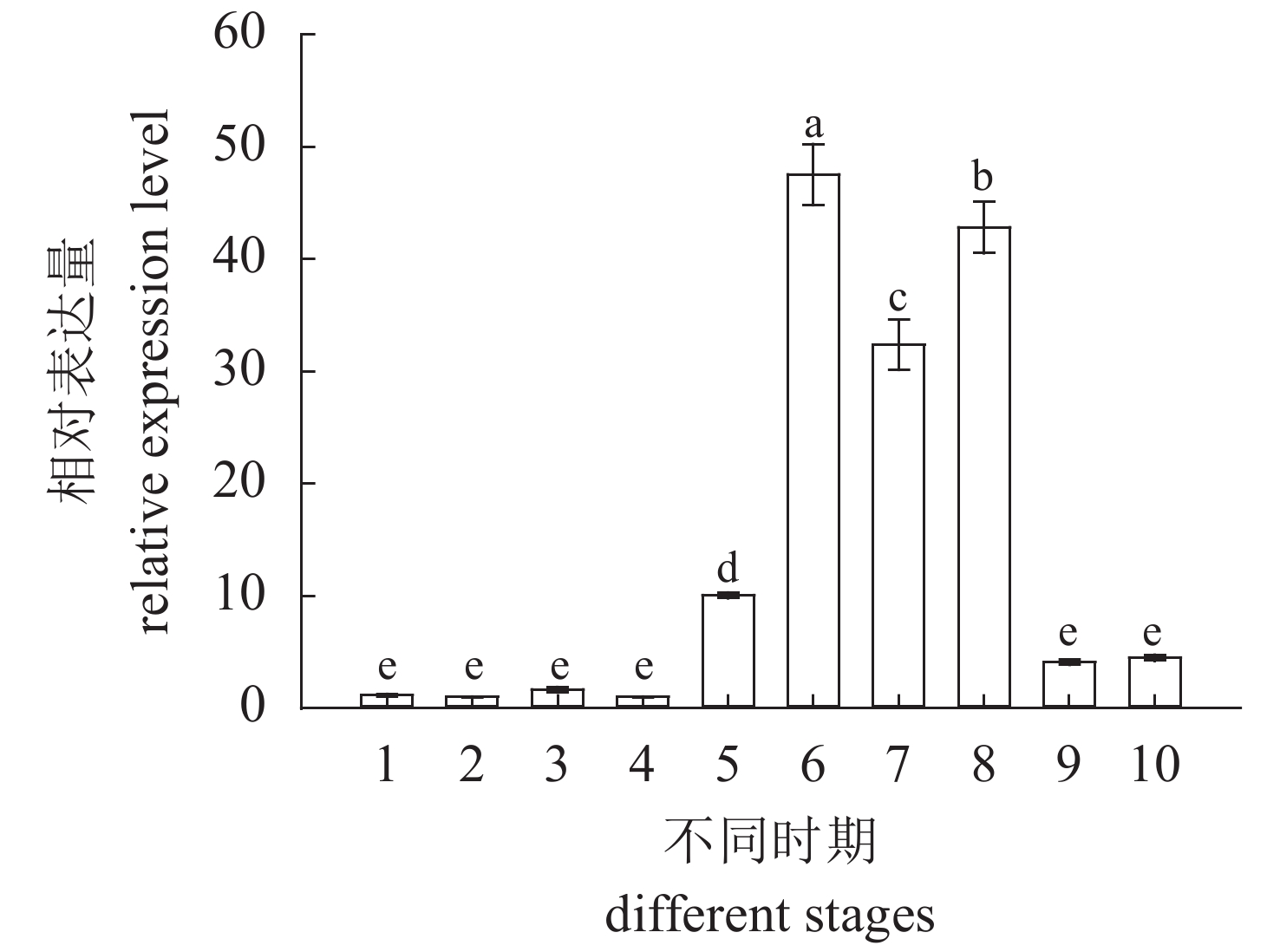
图 1 cbx2在青鳉胚胎发育中的定量表达分析
Figure 1. Real-time PCR analysis of cbx2 mRNA in different developmental stages of O. latipes embryos

图 2 青鳉cbx2在性腺中的定量表达
Figure 2. Real-time PCR of cbx2 mRNA in O. latipes gonads

图 图版Ⅱ 青鳉精巢和卵巢免疫组化(IHC)分析
Figure 图版Ⅱ. Immunohistochemistry (IHC) analysis in ovary and testis of O. latipes
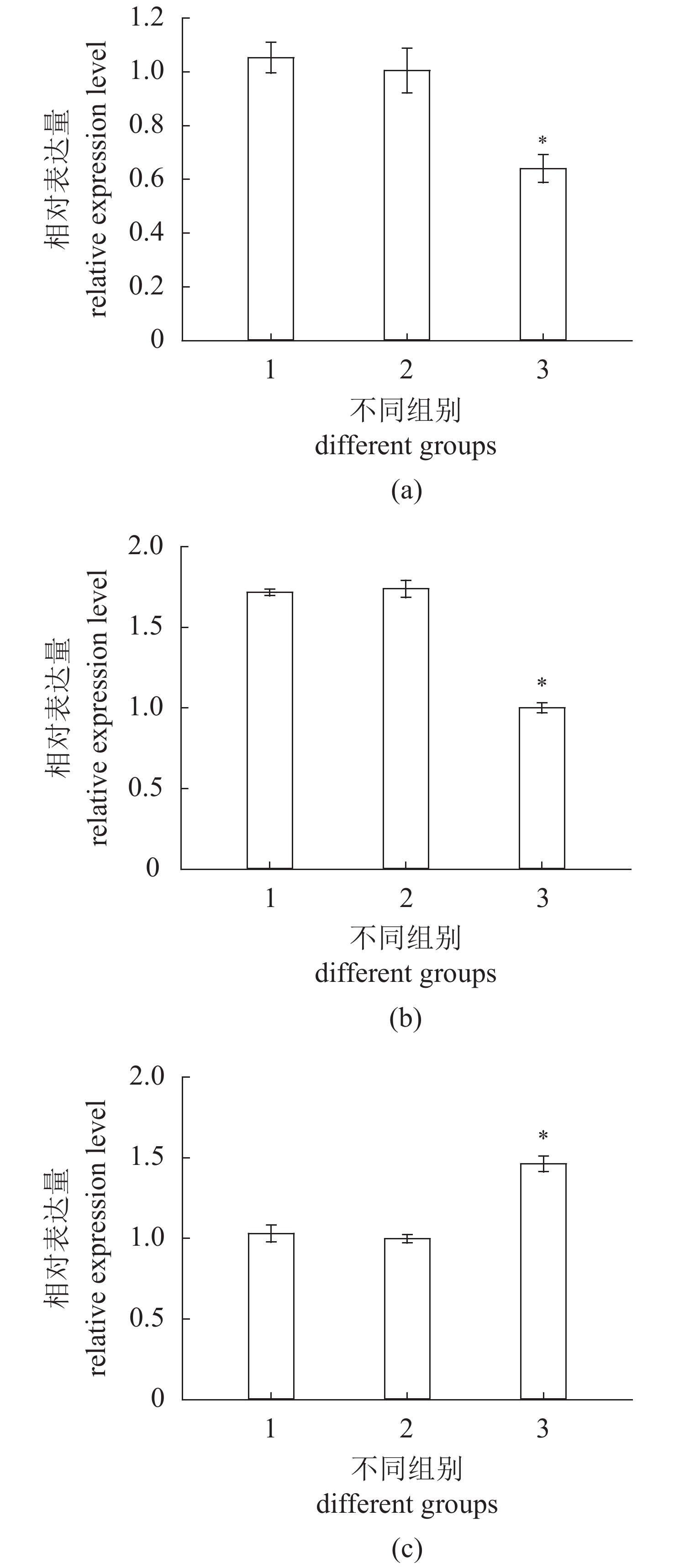
图 3 RNA干扰后cbx2(a)、sox9(b)和foxl2(c)的表达变化
Figure 3. Expression changes of cbx2(a), sox9(b) and foxl2(c) after RNA interference
表 1 Real-time PCR引物
Table 1. Primers used for Real-time PCR
引物
primer序列(5′→3′)
sequence (5′→3′)cbx2-F GCCCAAGTCCAGCACCTCA cbx2-R GCTCCTTCGCCTCACCTCT sox9-F AAACTGGCCGACCAATAC sox9-R CTCAGCCTCCTCCACAAA foxl2-F TCCTACACGTCCTGCCAGAT foxl2-R CCCATGCCGTTGTAAGAGTT 18S-F CTGAGAAACGGCTACCACAG 18S-R CAGCAACTTTAAGATACGC 表 2 siRNA靶位点和合成序列
Table 2. siRNA target site and sequence
基因
gene方向
direction序列
sequencecbx2-siRNA-1 (251) sense (5′-3′) CCGACUCUGACCGCACUAATT anti-sense (3′-5′) UUAGUGCGGUCAGAGUCGGTT cbx2-siRNA-2 (853) sense (5′-3′) GCGCUGCACCUGAACCCUUTT anti-sense (3′-5′) AAGGGUUCAGGUGCAGCGCTT cbx2-siRNA-3 (1337) sense (5′-3′) GCCUCAUCGAGCACGUGUUTT anti-sense (3′-5′) AACACGUGCUCGAUGAGGCTT -
Alkema M J, Bronk M, Verhoeven E, et al. Identification of bmi1-interacting proteins as constituents of a multimeric mammalian polycomb complex[J]. Genes & Development, 1997, 11(2): 226-240.
Bulyzhenkov V E, Ginter E K, Ivanov V I. Interaction of the homoeotic mutations aristapedia and polycomb during ontogenesis of Drosophila melanogaster[J]. Ontogenez, 1974, 5(6): 634-641.
Katoh-Fukui Y, Miyabayashi K, Komatsu T, et al. Cbx2, a polycomb group gene, is required for Sry gene expression in mice[J]. Endocrinology, 2012, 153(2): 913-924. doi: 10.1210/en.2011-1055
Biason-Lauber A, Konrad D, Meyer M, et al. Ovaries and female phenotype in a girl with 46, XY karyotype and mutations in the CBX2 gene[J]. The American Journal of Human Genetics, 2009, 84(5): 658-663. doi: 10.1016/j.ajhg.2009.03.016
Sproll P, Eid W, Biason-Lauber A. CBX2-dependent transcriptional landscape: implications for human sex development and its defects[J]. Scientific Reports, 2019, 9(1): 16552. doi: 10.1038/s41598-019-53006-7
Matsuda M, Nagahama Y, Shinomiya A, et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish[J]. Nature, 2002, 417(6888): 559-563. doi: 10.1038/nature751
Kang Y, Guan G J, Hong Y H. Insights of sex determination and differentiation from medaka as a teleost model[J]. Hereditas, 2017, 39(6): 441-454(in Chinese).
Iwamatsu T. Stages of normal development in the medaka Oryzias latipes[J]. Mechanisms of Development, 2004, 121(7-8): 605-618. doi: 10.1016/j.mod.2004.03.012
Arnold S J, Robertson E J. Making a commitment: cell lineage allocation and axis patterning in the early mouse embryo[J]. Nature Reviews Molecular Cell Biology, 2009, 10(2): 91-103. doi: 10.1038/nrm2618
Viotti M, Nowotschin S, Hadjantonakis A K. SOX17 links gut endoderm morphogenesis and germ layer segregation[J]. Nature Cell Biology, 2014, 16(12): 1146-1156. doi: 10.1038/ncb3070
Stower M J, Bertocchini F. The evolution of amniote gastrulation: the blastopore-primitive streak transition[J]. Wiley Interdisciplinary Reviews: Developmental Biology, 2017, 6(2): e262. doi: 10.1002/wdev.262
Zhou X L, Sasaki H, Lowe L, et al. Nodal is a novel TGF-β-like gene expressed in the mouse node during gastrulation[J]. Nature, 1993, 361(6412): 543-547. doi: 10.1038/361543a0
Yin H C, Lü H Y. Embryonic development and primordial germ cell migration in culter Ancherythroculter nigrocauda[J]. Fisheries Science, 2010, 29(5): 265-269(in Chinese). doi: 10.3969/j.issn.1003-1111.2010.05.004
Wang X Y, Zhang J L, Shi Z Y. Molecular characterization and tissue expression of cbx2 gene in Paralichthys olivaceus[J]. Biotechnology Bulletin, 2019, 35(4): 69-75(in Chinese).
Eid W, Opitz L, Biason-Lauber A. Genome-wide identification of CBX2 targets: insights in the human sex development network[J]. Molecular Endocrinology, 2015, 29(2): 247-257. doi: 10.1210/me.2014-1339
Noguchi K, Shiurba R, Higashinakagawa T. Nuclear translocation of mouse polycomb M33 protein in regenerating liver[J]. Biochemical and Biophysical Research Communications, 2002, 291(3): 508-515. doi: 10.1006/bbrc.2002.6480
Hirose S, Komoike Y, Higashinakagawa T. Identification of a nuclear localization signal in mouse polycomb protein, M33[J]. Zoological Science, 2006, 23(9): 785-791. doi: 10.2108/zsj.23.785
Baumann C, De La Fuente R. Role of polycomb group protein Cbx2/M33 in meiosis onset and maintenance of chromosome stability in the mammalian germline[J]. Genes, 2011, 2(1): 59-80. doi: 10.3390/genes2010059
Wang D S, Kobayashi T, Zhou L Y, et al. Foxl2 up-regulates aromatase gene transcription in a female-specific manner by binding to the promoter as well as interacting with ad4 binding protein/steroidogenic factor 1[J]. Molecular Endocrinology, 2007, 21(3): 712-725. doi: 10.1210/me.2006-0248
Suzuki H, Kanai-Azuma M, Kanai Y. From sex determination to initial folliculogenesis in mammalian ovaries: morphogenetic waves along the anteroposterior and dorsoventral axes[J]. Sexual Development, 2015, 9(4): 190-204. doi: 10.1159/000440689
Sekido R, Lovell-Badge R. Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer[J]. Nature, 2008, 453(7197): 930-934. doi: 10.1038/nature06944
Kim Y, Kobayashi A, Sekido R, et al. Fgf9 and Wnt4 act as antagonistic signals to regulate mammalian sex determination[J]. PLoS Biology, 2006, 4(6): e187. doi: 10.1371/journal.pbio.0040187
Chiang E FL, Pai C I, Wyatt M, et al. Two Sox9 genes on duplicated zebrafish chromosomes: expression of similar transcription activators in distinct sites[J]. Developmental Biology, 2001, 231(1): 149-163. doi: 10.1006/dbio.2000.0129
Zhang M, Zhu Y Y, Li W B, et al. Cloning and expression of sox9a/b gene in the large yellow croaker (Larimichthys crocea)[J]. Journal of Fisheries of China, 2019, 43(8): 1691-1705(in Chinese).
Katoh-Fukui Y, Owaki A, Toyama Y, et al. Mouse polycomb M33 is required for splenic vascular and adrenal gland formation through regulating Ad4BP/SF1 expression[J]. Blood, 2005, 106(5): 1612-1620. doi: 10.1182/blood-2004-08-3367

图(5)
表(2)
计量
- 文章访问数: 509
- PDF下载数: 85
- 施引文献: 0


 下载:
全尺寸图片
幻灯片
下载:
全尺寸图片
幻灯片

