- 1. MEASUREMENT
A simple
definition of
physics is “the
science of
measurement”
- 2. Measuring-
For a measurement
to be useful it must:
– be accurate
– have a unit.
- 3. Basic measurements
in physics
• Length:- Including areas
and volumes
• Mass
• Time
• Temperature etc.
- 4. Time
Time allows change to happen.
Measuring instruments:
• Stop watch.
• Electronic timer (for very short
times).
Units
seconds (s), minutes (m) and hours
(h or hrs) (multiply each time by 60 to
use the smaller measure)
- 5. Temperature
Temperature measures how
hot or cold an object is.
Measuring instrument:
• Thermometer.
Unit
degrees Celsius (0C)
Degree Fahrenheit (0F)
- 6. Length
Measuring instruments:
• Tape measure
• Metre stick
• Opisometer (for small curves)
• Trundle wheel (for longer distances)
Units
metres (m) or centimetres (cm)
- 7. Opisometer
(for small curves)
• most commonly used to
measure the lengths of roads,
rivers and other line features
on maps.
• Opisometers designed for this
purpose provide scales reading
the measured distance in
- 8. The trundle wheel
• a simplified form of surveyor's wheel
• commonly used by school children
• an easy way to find the rough
distance
• The trundle a clicking device which is
triggered once per revolution of the
wheel
• are not as accurate as other methods
• but are a good way to get a rough
estimation of a fairly long distance
over a good surface.
- 9. Mass
DEFINITION
Mass- the amount of matter an
object has
Matter- something that has mass
and takes up space.
- 10. Measuring Devices of Mass
- 11. Weights
- 12. Units for Measuring Mass
• SI Unit- Kilogram (kg) or gram (g)
• 1 paper clip= 1 gram
• 1000g=1 kilogram
• The average human adult weighs 75kg.
• Mass cannot be changed by the
location, shape and speed of the body.
• Large masses are measured in tonnes
while small masses are measured in
grams.
- 13. Examples of Masses of objects
Mass in
Object
kilogram (kg)
Electron 10-30
A fine grain of sand 10-6
A pea 10-3
An apple 10-1
A medium-sized car 103
Earth 1024
Sun 1030
- 14. Weight
Spring Definition- Weight is a
gravitational force acting on
balance an object
Formula- W = mg
where m is the mass of
the object
g is the gravitational force
Units- Weight is measured in
newtons (N)
- 15. Gravitational Field
The gravitational field is a region
in which a mass experiences a
force due to gravitational
attraction.
- 16. Weight..???
Mass..???
I am totally
confused!
- 17. Pretend That You Are in space!
Oh where, oh where,
has my weight gone?
Oh where, oh where
can it be?
You are floating around.
You drift over to a
floating scale and put your
feet on it. Your feet do
not push down on the scale
at all. The scale shows
that you weigh 0. You
have lost your weight—but
did you lose your mass?
- 18. All my body parts are
still together — so I
still have mass!
- 19. On a space flight, your
mass would be the same;
but your weight would
change.
- 20. Summing up
Mass and Weight
• Your MASS is the amount of
material in your body.
• Your MASS doesn’t depend on
where you are.
• Your WEIGHT is how much your
body pushes down on a scale.
• Your WEIGHT depends on how
much MASS you have and where
you are.
- 21. Where would you weigh the
most?
Earth
OR
Moon
- 22. Remember,
• Gravity describes the pull between
masses.
• You have mass, and the earth has
mass.
• There is a pull between you and
the earth.
• We measure this pull with a scale.
• The more the gravity, the greater
the squeeze on the springs of the
scale…and the more you weigh.
- 23. Gravity and Weight
Summary
• Your weight on earth is a
measure of the GRAVITY pull
between you and the earth.
• Bodies with more MASS
produce a greater GRAVITY
pull on each other.
- 24. On Earth
So, if I ate a lot
of yummy tuna fish
while standing on
the scale, I would
gain mass.
The pull between
the earth and me
would become
greater—and I
would weigh more!
- 25. Back to the Moon
During my trip to
the moon, my mass
does not change,
but the moon has
much less mass than
the earth.
- 26. Still on the Moon!
There is less gravity pull
between you and the moon.
If you stood on the scale on
the moon—you would weigh
less than on earth!
- 27. Differences between
Weight and Mass
Weight Mass
• pull of gravity on • amount of matter in
the body the body
• units are newtons • units are kilograms,
& dyne grams
• changes from place • is constant regardless
to place of place or location
• measure with the • measure with the
help of spring help of beam balance
balance
- 28. VOLUME:
What Is Volume?
Space occupied by an
object is known as its
volume.
It is denoted by ‘V’
- 29. Units Of Volume
Volume is measured in
cubic centimetres.
Also called centimetre cube
(cm3)
Bigger unit of volume is
cubic metre or meter cube
(m3)
- 30. Volume of Liquids
Volume of liquid is measured
using measuring vessels.
Its unit is litre and smaller
unit is millilitre.
1 litre = 1000 ml
1 litre = 1000 cubic cm
So,
1 ml = 1 cubic cm
- 31. Different types of measuring
vessels
- 32. How to measure VOLUME
of liquids
We will be using
graduated cylinders to
find the volume of liquids
and other objects.
Read the measurement based on
the bottom of the meniscus or
curve. When using a real cylinder,
make sure you are eye-level with
the level of the water.
What is the volume of water in the
cylinder? _____mL
- 37. What causes the meniscus?
A concave meniscus occurs when the
molecules of the liquid attract those
of the container.The glass attracts
the water on the sides.
- 38. VOLUME of solids having
regular geometric shapes
Volume of solids having regular
geometric shapes can be
determined using different
formulae.
- 39. Cube
Formula to find volume of a
cube is- L x L x L = L3
So,volume of the cube
= 1cm X 1cm X 1cm
= 1 cm3
- 40. Cuboid
Height (H)
- 41. Volume of Cuboids.
Look at the
adjacent
cuboid:
We must first calculate the area of the
base of the cuboid:
The base is a rectangle measuring 10cm
by 3cm:
10cm
3cm
- 42. 10cm
3cm
Area of a rectangle = length x width
Area = 10 x 3
Area = 30cm2
We can place 30 cubic centimetres on the base:
- 43. 4cm
3cm
10cm
We have now got to find how many layers
of 1cm cubes we can place in the cuboid:
We can fit in 4 layers.
Volume = 30 x 4
Volume = 120cm3
That means that we can place 120 of our
cubes measuring a centimetre in all directions
inside our cuboid.
- 44. 4cm
3cm
10cm
We have found that the volume of the cuboid is given
by:
Volume = 10 x 3 x 4 = 120cm3
This gives us our formula for the volume of a cuboid:
Volume = Length x Width x Height
- 45. The diagram shows a cuboid.
Work out the volume, in cm3, of
the cuboid.
- 46. Volume of Irregular Objects
• For example: a rock!
• When objects do not take on
a regular shape or it is
difficult to measure their
dimensions we use a different
method to find their volume,
this method is called water
displacement
- 47. Displacement is:
– The amount of water an
object replaces is equal to
the object’s volume
How to use the method
of water displacement?
- 48. Measuring the volume of an
irregularly shaped object that
sinks in water
Graduated
cylinder
Stone
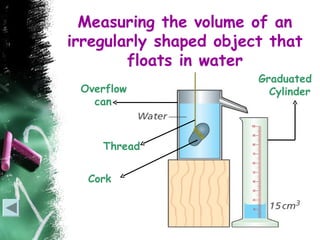
- 49. Measuring the volume of an
irregularly shaped object that
floats in water
Graduated
Overflow Cylinder
can
Thread
Cork
- 50. Story of a naked man
In the first century BC the Roman
architect Vitruvius related a story of how
Archimedes uncovered a fraud in the
manufacture of a golden crown commissioned
by Hiero II, the king of Syracuse. The crown
(corona in Vitruvius’s Latin) would have been in
the form of a wreath. Hiero would have placed
such a wreath on the statue of a god or
goddess. Suspecting that the goldsmith might
have replaced some of the gold given to him by
an equal weight of silver, Hiero asked
Archimedes to determine whether the wreath
was pure gold. And because the wreath was a
holy object dedicated to the gods, he could
not disturb the wreath in any way.
- 51. Archimedes
The solution which occurred when he
stepped into his bath and caused it to
overflow was to put a weight of gold
equal to the crown, and known to be
pure, into a bowl which was filled with
water to the brim. Then the gold would
be removed and the king’s crown put in,
in its place. An alloy of lighter silver
would increase the bulk of the crown
and cause the bowl to overflow.
- 52. Does something that is
bigger always have more
mass?
- 53. Does bigger = more mass?
- 54. What is density?
• Density is a comparison of how much
matter there is in a certain amount
of space.
Definition- Density is defined as
mass per unit volume
Formula- mass/volume
S I units- kg/m 3 M
Other units- g/cm3
D V
My Dear Valentine
- 55. Density
Density Sink
Object Mass/g Vol/cm 3
or
g/cm3
Float?
Aluminium
50 18.52 2.70 Sink
sheet
Gold
150 7.89 19.0 Sink
bracelet
Water 500 500 1 ---
Ice 10 10.87 0.92 Float
Block of 0.5 Float
800 1600
pinewood
- 56. Which one is more dense?
- 57. Now which one is
more dense?
- 58. Ways to Affect Density
• Change mass AND keep
volume same
• Change volume AND
keep mass same
- 59. Change Mass AND
Keep Volume Same
• Increase the mass increase density
• Decrease the mass decrease in density
Which container has more density?
A B
- 60. Change Volume AND
Keep Mass Same
• Increase the volume decrease density
• Decrease the volume increase density
• Which container has more density?
A B
- 61. What 2 ways will increase density?
Keep the
same mass
AND
decrease
the volume
Keep the
same volume
AND
increase the
mass